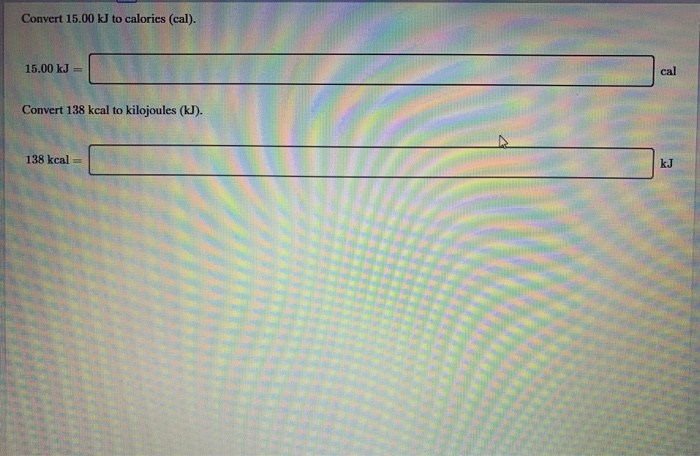
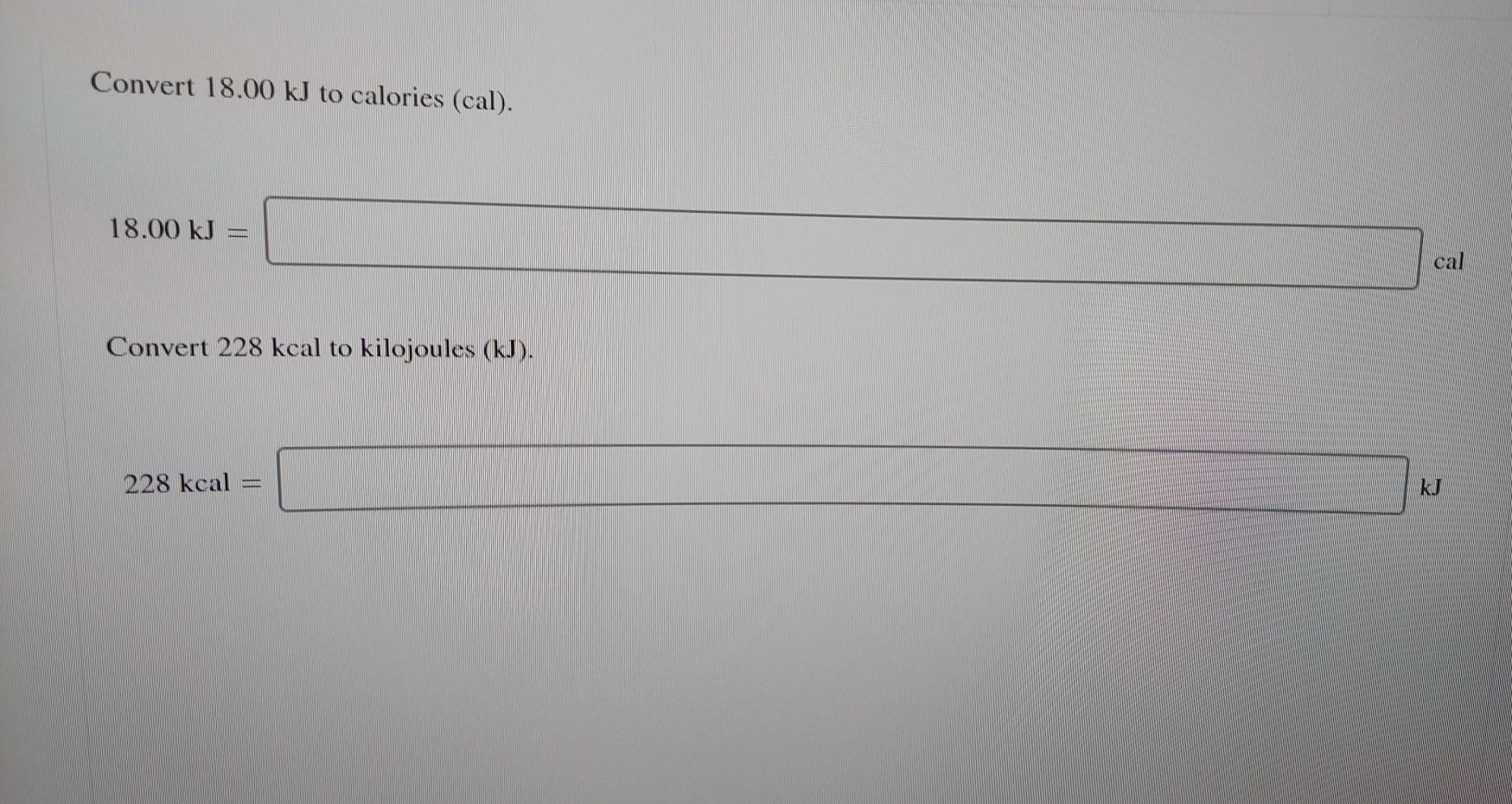
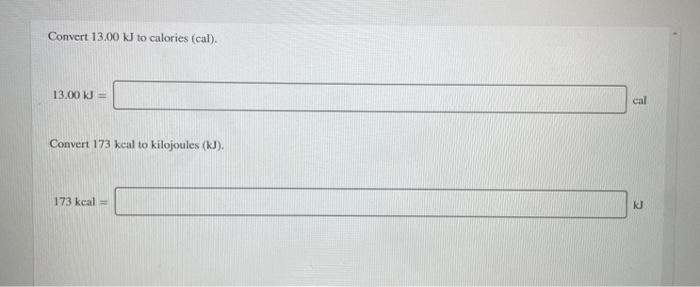
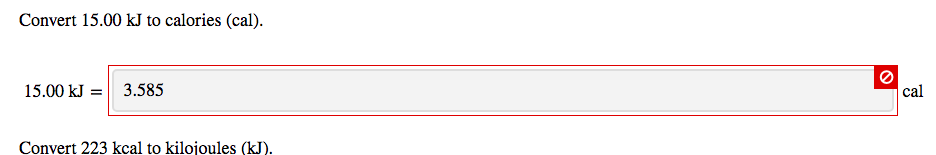
SOLVED:Perform the indicated conversions. a. 91.74 kcal into calories b. 1.781 kJ into calories c. 4.318 \times 10^{3} \mathrm{J} into kilocalories d. 9.173 \times 10^{4} cal into kilojoules

SOLVED:Perform the indicated conversions. a. 91.74 kcal into calories b. 1.781 \mathrm{~kJ} into calories c. 4.318 \times 10^{3} J into kilocalories d. 9.173 \times 10^{4} cal into kilojoules

SOLVED:Convert the following numbers of calories or kilocalories into joules and kilojoules (Remember: Kilo means 1000.) \begin{array}{ll}{\text { a. } 75.2 \mathrm{kcal}} & {\text { c. } 1.41 \times 10^{3} \mathrm{cal}} \\ {\